The Research Network
Functional Nanostructures
was funded by the
Baden-Württemberg Stiftung.
Ordered superstructures in nanometer scale of macromolecules like proteins, polysaccharides and inorganic nanoparticles possess excellent physical properties as the biocomposites such as bones, and nacre already showed. However, the directed connection of those organic and inorganic components in the range of few nanometers is still challenging. The goal of our project is the self-assembly of nanoparticles by using two proteins with incorporated peptide sequence for the non-classical crystallization of nanoparticles into defined structures with perfect alignment of the building units. The donut-like, fiber forming protein Hcp1 with thiol functionality is supposed to direct the self-assembly of nanoparticles into fiber structures. Furthermore 3-D structures of nanoparticles can be realized by applying Hcp1 protein as scaffold and the elastin like protein (ELP) for nanoparticle binding and linkage with the scaffold. The ELP can be modified with different functional groups or peptide sequences for binding of several nanoparticles. For our modular strategy we intend to choose biominerals such as calcium carbonate, -phosphate, oxalate and metallic nanoparticles (Au, Ag), semiconductor (CdS, ZnS) and magnetic nanoparticles (Fe3O4). Our strategy of the non-classical crystallization can provide hybrid materials with optical, magnetic and mechanical properties. It will allow us to produce for example nanoscopic, elastic, switchable magnets, which can be utilized in electronic data storage, medical diagnostics such as contrast agent or in sensor devices.
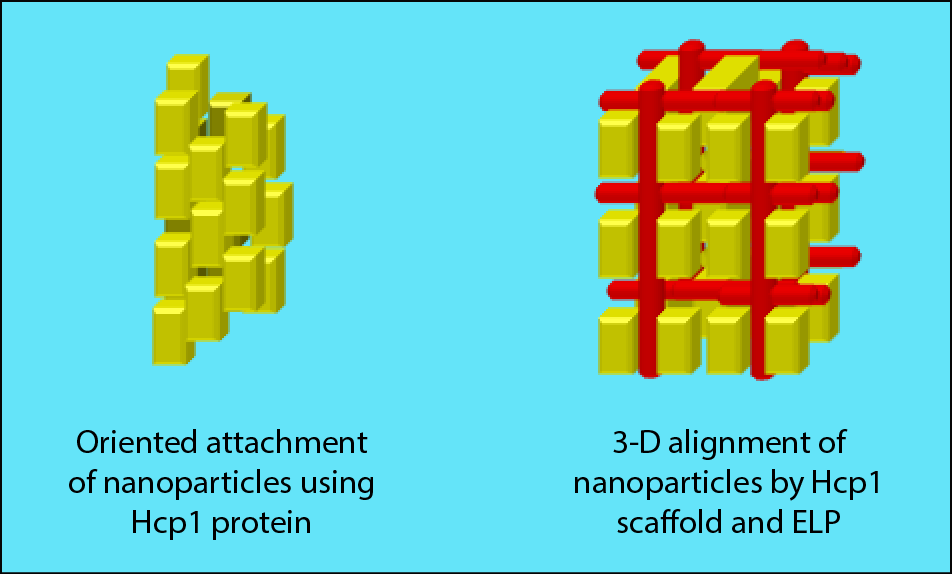
Fig.1: Scheme for the ordered structure formation of nanoparticles (yellow symbol).